  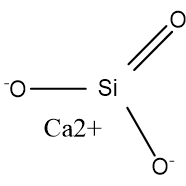
In particular, the reaction is not observed with suspensions of colloidal silica. In a typical preparation, monomeric orthosilicate was found to react completely in 75 seconds dimeric pyrosilicate in 10 minutes and higher oligomers in considerably longer time. Silicate anions in solution react with molybdate anions yielding yellow silicomolybdate complexes. Some plants excrete ligands that dissolve silicates, a step in biomineralization. Such equilibria are relevant to the processes occurring on geological time scales. The chief challenge is the very low solubility of SiO 4 4- and its various protonated forms. When treated with calcium oxides and water, silicate minerals form Portland cement.Įquilibria involving hydrolysis of silicate minerals are difficult to study. Their resiliency also recommends their use as building materials. Silicates are generally inert chemically. Silicates of non-alkali cations, or with sheet and tridimensional polymeric anions, generally have negligible solubility in water at normal conditions. Soluble sodium silicates and mixtures thereof, known as waterglass are in fact important industrial and household chemicals. They form several solid hydrates when crystallized from solution. Silicates with alkali cations and small or chain-like anions, such as sodium ortho- and metasilicate, are fairly soluble in water. Īt very high pressure, such as exists in the majority of the earth's crust, even SiO 2 adopts the six-coordinated octahedral geometry in the mineral stishovite, a dense polymorph of silica found in the lower mantle of the Earth and also formed by shock during meteorite impacts. This structure is also seen in the hexahydroxysilicate anion Si(OH) 2−Ħ that occurs in thaumasite, a mineral found rarely in nature but sometimes observed among other calcium silicate hydrates artificially formed in cement and concrete submitted to a severe sulfate attack. For example, in the anion hexafluorosilicate SiF 2−Ħ, the silicon atom is surrounded by six fluorine atoms in an octahedral arrangement. Silicates with non-tetrahedral silicon Īlthough the tetrahedron is a common coordination geometry for silicon(IV) compounds, silicon may also occur with higher coordination numbers. In a framework silicate, known as a tectosilicate, each tetrahedron shares all 4 oxygen atoms with its neighbours, forming a 3D structure. Both muscovite and biotite have very weak layers that can be peeled off in sheets. This structure does lead to minerals in this group having one strong cleavage plane. In this group, known as phyllosilicates, tetrahedra all share three oxygen atoms each and in turn link to form two-dimensional sheets. Two or more silicon atoms can share oxygen atoms in various ways, to form more complex anions, such as pyrosilicate Si A common mineral in this group is olivine ( (Mg,Fe) 2SiO 4). Isolated orthosilicate anions have the formula SiO 4−Ĥ. The silicates can be classified according to the length and crosslinking of the silicate anions. This Si-O-M-O-Si linkage is strong and rigid, which properties are manifested in the rock-like silicates. The oxygen atoms, which bears some negative charge, link to other cations (M n+). In all silicates, silicon atom occupies the center of an idealized tetrahedron whose corners are four oxygen atoms, connected to it by single covalent bonds according to the octet rule. The name "silicate" is sometimes extended to any anions containing silicon, even if they do not fit the general formula or contain other atoms besides oxygen such as hexafluorosilicate 2−.Most commonly, silicates are encountered as silicate minerals.įor diverse manufacturing, technological, and artistic needs, silicates are versatile materials, both natural (such as granite, gravel, and garnet) and artificial (such as Portland cement, ceramics, glass, and waterglass). The name is also used for any salt of such anions, such as sodium metasilicate or any ester containing the corresponding chemical group, such as tetramethyl orthosilicate. The family includes orthosilicate SiO 4− 4 ( x = 0), metasilicate SiO 2− 3 ( x = 1), and pyrosilicate Si 2O 6− 7 ( x = 0.5, n = 2). In chemistry, a silicate is any member of a family of polyatomic anions consisting of silicon and oxygen, usually with the general formula [SiO (4-2 x)− Structure of the orthosilicate anion SiO 4− 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
